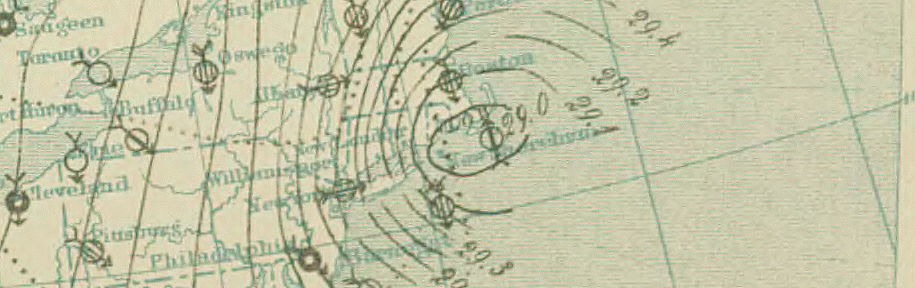
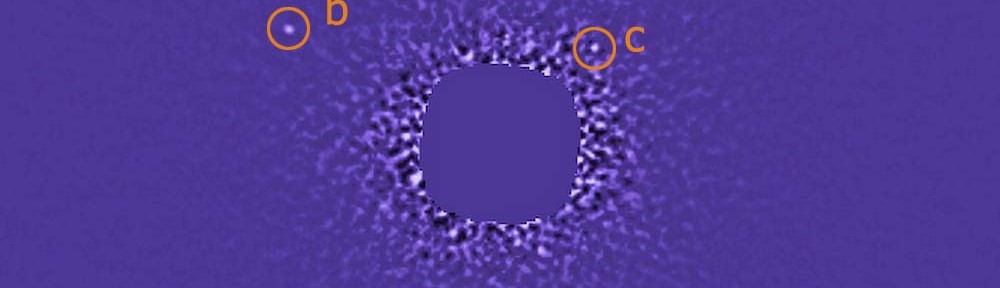
Last time, we looked at the agony and ecstasy of the life of Robert FitzRoy, the world’s first weather forecaster. Like most other weather forecasters, FitzRoy provided an invaluable service, and yet was mocked resoundingly when his forecasts were in error. Unlike most weather forecasters, this criticism drove him deeper into an already deep depression, and he ended up committing suicide only a few years into his new gig. We may empathize with FitzRoy’s plight, but who among us hasn’t at some point been in awe of the sheer inaccuracy of a weather forecast? Why should FitzRoy have it easier than Lady Catherine, Jane Austen’s archetypal ultrarich know-nothing? Of course, this naturally leads us to the question: will we ever be able to make a truly accurate weather forecast? The answer to this question is a big fat no. But the path to that big fat no is quite a bit more interesting than you might expect.